
Sarvarthapedia is a comprehensive collection of universal knowledge
Atomic Theory, Periodic Table, Chemical Reactions and Molecular Basis of Life and Consciousness
Chemistry is the scientific study of matter, its composition, structure, properties, and the transformations it undergoes, forming a foundational discipline that bridges the physical sciences and the life sciences while providing a unified framework for understanding the material universe. At its most fundamental level, chemistry is concerned with atomic structure, which describes how matter is composed of atoms, themselves consisting of protons, neutrons, and electrons arranged in defined configurations governed by the principles of quantum mechanics. The development of atomic theory owes much to pioneering figures such as John Dalton, Niels Bohr, and Erwin Schrödinger, whose models and equations helped establish a deeper understanding of electron orbitals, energy levels, and probabilistic distributions that define chemical behavior.
Periodic table
The organization of elements into the periodic table represents one of the most powerful conceptual tools in chemistry, allowing scientists to predict properties and reactivity based on atomic number and electron configuration. The periodic system, first systematically arranged by Dmitri Mendeleev, reveals recurring patterns known as periodicity, which govern trends such as atomic radius, ionization energy, and electronegativity. These periodic relationships underpin the nature of chemical bonding, including ionic bonds, covalent bonds, and metallic bonds, which in turn determine the formation and stability of compounds.
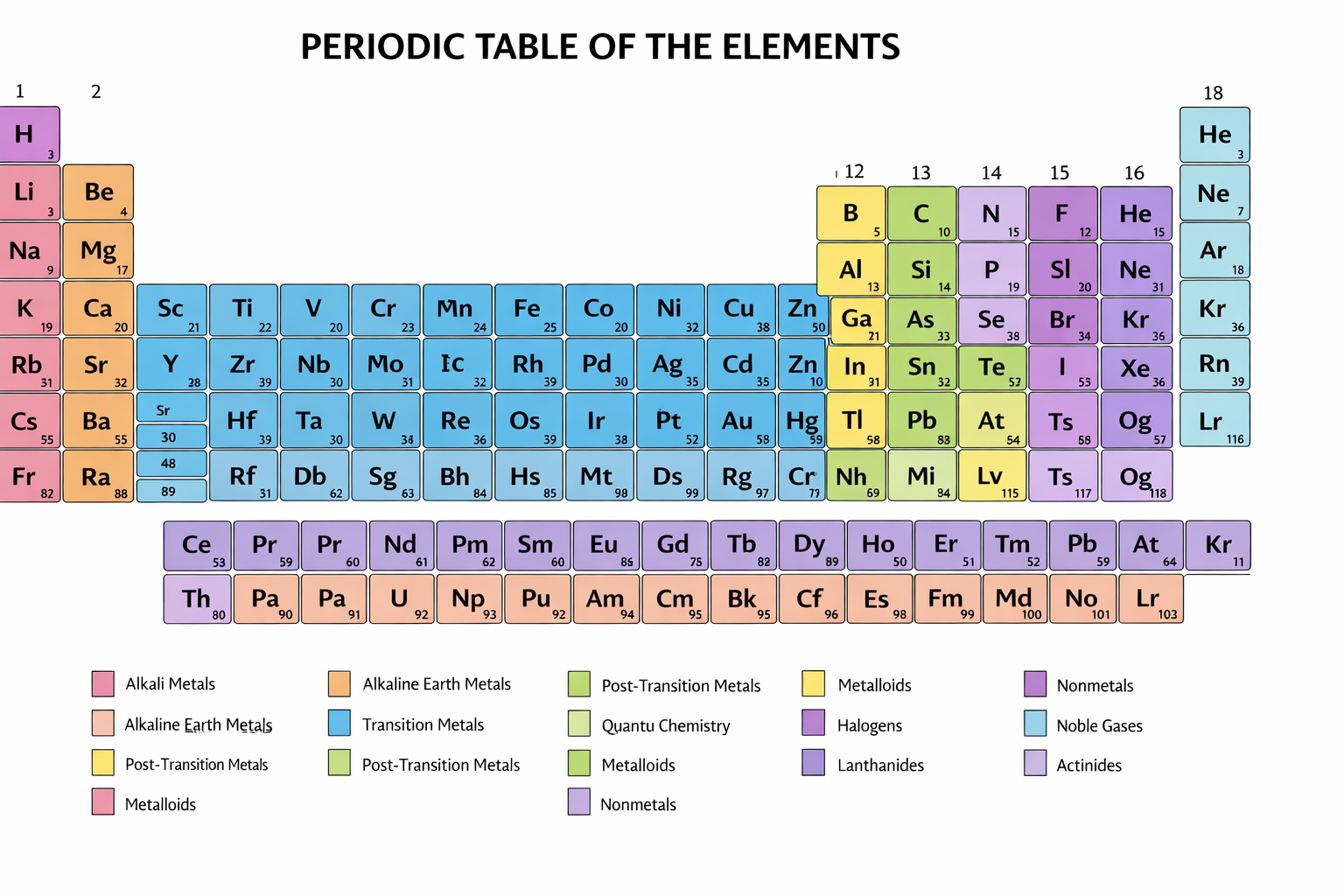
Central to chemistry is the study of chemical reactions, processes in which substances transform into new substances through the rearrangement of atoms and the making and breaking of chemical bonds. These reactions obey fundamental laws such as the law of conservation of mass and are influenced by factors including temperature, pressure, concentration, and the presence of catalysts. Reaction types such as acid-base reactions, redox reactions, and precipitation reactions illustrate the diversity of chemical transformations. The field of chemical kinetics examines the rates of reactions, while thermodynamics explores the energy changes involved, including concepts such as enthalpy, entropy, and Gibbs free energy.
Chemistry is traditionally divided into several major branches, including physical chemistry, organic chemistry, and materials chemistry, each addressing different aspects of matter and its transformations. Physical chemistry applies the principles of physics to chemical systems, focusing on energy transfer, molecular motion, and quantum phenomena. Organic chemistry centers on the study of carbon-based compounds, which form the basis of life and include vast classes of molecules such as hydrocarbons, polymers, and pharmaceuticals. The work of Friedrich Wöhler, who synthesized urea from inorganic compounds, marked a turning point in organic chemistry by demonstrating that organic molecules could be created artificially. Materials chemistry, meanwhile, focuses on the design and synthesis of substances with specific properties, including nanomaterials, semiconductors, and biomaterials, which have applications in technology, medicine, and industry.
A closely related field is biochemistry, which explores the chemical processes within living organisms, revealing that life itself is fundamentally a network of chemical reactions. Within this domain, nucleic acids such as DNA and RNA play a central role in storing and transmitting genetic information, enabling the processes of replication, transcription, and translation that sustain life. The elucidation of the double helix structure of DNA by James Watson and Francis Crick, building on the work of Rosalind Franklin, revolutionized biology and demonstrated the profound chemical basis of heredity. Proteins, enzymes, lipids, and carbohydrates further illustrate how complex biological systems emerge from relatively simple chemical building blocks.
From a philosophical and scientific perspective, human beings themselves can be understood as highly organized assemblies of chemical compounds, structured into cells, tissues, and organs through intricate biochemical pathways. In this view, life and the concept of a life force are not separate metaphysical entities but rather emergent properties arising from the dynamic interactions of molecules. The processes of metabolism, homeostasis, and signal transduction reflect the continuous flow and transformation of chemical substances within the body. Even events such as an accidental death can be interpreted through the lens of chemistry, as the outcome of complex interactions between biological systems and external conditions, governed by the deterministic and probabilistic behavior of molecular systems, suggesting that what appears random may in fact be rooted in underlying chemical processes.
Chemistry is deeply embedded in the history and structure of the universe itself. At the moment of the Big Bang, the universe consisted primarily of simple particles that eventually formed the first atoms, predominantly hydrogen and helium. Through processes of nuclear fusion within stars such as the Sun, heavier elements were synthesized, giving rise to the diversity of matter observed today. The formation of planets involved the aggregation of these elements into complex structures, with chemical differentiation leading to the development of atmospheres, oceans, and geological features. Thus, chemistry provides a continuous narrative from the origins of the universe to the formation of planetary systems and the emergence of life.
The advancement of chemistry has been closely associated with leading institutions and centers of research, including Massachusetts Institute of Technology, University of Cambridge, Harvard University, and Indian Institute of Science, where groundbreaking discoveries continue to shape the field. These institutions foster interdisciplinary collaboration, integrating chemistry with physics, biology, engineering, and computational sciences to address complex global challenges.
The commercial use of chemistry is vast and pervasive, encompassing industries such as pharmaceuticals, agriculture, energy, manufacturing, and consumer goods. Chemical processes are essential for the production of medicines, fertilizers, plastics, fuels, and countless other products that sustain modern life. The development of green chemistry aims to minimize environmental impact by designing processes that reduce waste and avoid hazardous substances, reflecting a growing awareness of sustainability.
In contrast, the military use of chemistry has historically included the development of explosives, propellants, and chemical weapons, raising significant ethical and humanitarian concerns. The use of chemical agents in conflicts has led to international efforts to regulate and prohibit such practices, highlighting the dual-use nature of chemical knowledge.
The integration of chemistry with technology has given rise to innovations such as computational chemistry, artificial intelligence in chemical research, and nanotechnology, enabling scientists to model complex systems and design new materials with unprecedented precision. These developments underscore the evolving nature of chemistry as both a theoretical and applied science.
Indian chemistry
Indian chemistry represents a continuous and richly layered tradition extending from ancient philosophical speculation to modern scientific research. In Vedic India, early chemical knowledge was embedded in disciplines such as Ayurveda, where scholars like Charaka and Sushruta developed sophisticated understandings of herbal compounds, metallurgy, and pharmaceutical preparations, while texts of Rasashastra explored alchemy, mineral processing, and metal extraction, often linked with spiritual transformation. The classical and medieval periods saw advancements in zinc distillation, steel production (notably Wootz steel), and dyeing techniques, demonstrating practical chemical expertise.
In the modern era, Indian chemistry transitioned into a formal scientific discipline under the influence of global science, with pioneering contributions from figures such as Prafulla Chandra Ray, regarded as the father of modern Indian chemistry, and C. V. Raman, whose work on light scattering had chemical implications. Contemporary Indian chemists, working in institutions like the Indian Institute of Science and Indian Institutes of Technology, contribute to fields such as materials chemistry, biochemistry, and nanotechnology, integrating traditional knowledge with cutting-edge research, thereby positioning India as a significant participant in global chemical science.
Ultimately, chemistry provides a comprehensive framework for understanding the material world, from the smallest subatomic particles to the vast structures of the cosmos. It reveals the interconnectedness of all things, demonstrating that the diversity of matter and the complexity of life arise from the same fundamental principles.
Chemistry, from ancient alchemy up to 2026
Volume 1: History of Chemistry
1. Pre‑Chemistry & Ancient Practices (Before 500 BCE)
- Early metallurgy – Copper smelting (c. 5000 BCE), bronze (tin + copper, c. 3000 BCE), iron smelting (c. 1200 BCE)
- Egyptian chemistry – Glassmaking, faience, dyes, mummification (natron, resins)
- Mesopotamian chemistry – Perfumery, soap‑making, metalworking
- Indian chemistry – Steel (wootz), zinc extraction, dyes (indigo), Ayurvedic preparations (rasashastra)
- Chinese alchemy – Gunpowder (c. 9th century CE), porcelain, paper, ink, early distillation
2. Greek & Hellenistic Alchemy (500 BCE – 500 CE)
- Four‑element theory (Empedocles, Aristotle) – Earth, water, air, fire + quintessence (aether)
- Atomism (Democritus, Leucippus) – Indivisible atoms (ἄτομος) moving in void
- Aristotle’s transmutation – Base metals can be changed into noble metals
- Alexandrian alchemy – Zosimos of Panopolis (c. 300 CE): earliest alchemical writings, “chrysopoeia” (gold‑making)
3. Islamic Golden Age (700 – 1400 CE)
- Jabir ibn Hayyan (Geber) – Father of early chemistry: distillation, crystallization, sublimation, filtration; discovered sulfuric acid (H₂SO₄), nitric acid (HNO₃), aqua regia (dissolves gold); Jabirian corpus
- Al‑Razi (Rhazes) – Classification of substances (animal, vegetable, mineral)
- Al‑Biruni – Specific gravity measurements
- Ibn Sina (Avicenna) – Rejected transmutation of metals
4. European Alchemy & Iatrochemistry (1200 – 1600)
- Albertus Magnus – Discovered arsenic, described chemical affinity
- Roger Bacon – Opus Majus, experimental method, gunpowder formula
- Paracelsus – “Alchemy’s purpose is not to make gold but medicines” (iatrochemistry), introduced laudanum, zinc, tria prima (salt, sulfur, mercury)
- Georg Bauer (Agricola) – De Re Metallica (1556), systematic mining & metallurgy
5. The Chemical Revolution (1600 – 1800)
- Jan Baptist van Helmont – Coined “gas” (from chaos), quantitative experiment (willow tree growth), discovered carbon dioxide
- Robert Boyle – The Sceptical Chymist (1661): defined element as simplest substance, Boyle’s law (PV = constant)
- Johann Joachim Becher & Georg Ernst Stahl – Phlogiston theory (combustion releases “phlogiston”)
- Joseph Black – Rediscovered CO₂ (“fixed air”), latent heat, magnesium
- Henry Cavendish – Discovered hydrogen (“inflammable air”), measured Earth’s density
- Joseph Priestley – Discovered oxygen (1774), nitrous oxide (“laughing gas”), soda water
- Carl Wilhelm Scheele – Discovered oxygen (independently), chlorine, glycerol, many organic acids
- Antoine Lavoisier – Father of modern chemistry: law of conservation of mass, identified oxygen’s role in combustion, debunked phlogiston, named hydrogen and oxygen, wrote Traité Élémentaire de Chimie (1789, first modern chemistry textbook), developed metric system
- Joseph Proust – Law of definite proportions (same compound always same element ratios)
- John Dalton – Atomic theory (1803): all matter made of atoms, atoms of same element identical, compounds from fixed ratios (law of multiple proportions), first atomic weights
6. 19th Century: The Rise of Modern Chemistry (1800 – 1900)
- Jöns Jacob Berzelius – Developed chemical notation (letters for elements), measured atomic weights, discovered silicon, selenium, thorium, coined “catalysis”, “allotrope”, “polymer”
- Amedeo Avogadro – Avogadro’s law (equal volumes of gases contain equal number of molecules), Avogadro’s number (N_A)
- Friedrich Wöhler – Synthesized urea (1828, disproved vitalism), isolated aluminum
- Justus von Liebig – Organic analysis (combustion), Liebig condenser, agricultural chemistry (fertilizers)
- Louis Pasteur – Chirality (separated tartaric acid enantiomers), pasteurization, germ theory
- Dmitri Mendeleev & Lothar Meyer – Periodic table (1869): Mendeleev predicted undiscovered elements (eka‑aluminum → gallium, eka‑silicon → germanium)
- Jacobus van ’t Hoff – Stereochemistry (tetrahedral carbon, chiral centers), chemical kinetics, osmotic pressure, first Nobel Prize in Chemistry (1901)
- Svante Arrhenius – Electrolytic dissociation theory (ions), Arrhenius equation (temperature dependence of reaction rates)
- Wilhelm Ostwald – Catalysis, physical chemistry, founded physical chemistry journal
- Henri Becquerel, Marie & Pierre Curie – Radioactivity (uranium, polonium, radium)
- William Ramsay – Noble gases (He, Ne, Ar, Kr, Xe)
- J.J. Thomson – Discovery of electron (1897, cathode rays)
- Emil Fischer – Fischer projection, Fischer esterification, Fischer indole synthesis, sugar & purine chemistry (caffeine, uric acid)
7. 20th Century: Quantum & Molecular Revolution (1900 – 2000)
- Max Planck – Quantum hypothesis (1900, E = hν)
- Ernest Rutherford – Nuclear atom (gold foil experiment, 1909), discovered proton (1919)
- Niels Bohr – Bohr model of hydrogen (1913)
- Gilbert N. Lewis – Lewis dot structures, covalent bond, acid‑base theory (Lewis acids/bases), thermodynamics activity
- Linus Pauling – Electronegativity scale (Pauling scale), hybridization (sp, sp², sp³), resonance theory, α‑helix & β‑sheet (protein structure), Nobel Prizes in Chemistry (1954) & Peace (1962)
- Erwin Schrödinger & Werner Heisenberg – Quantum mechanics applied to atoms (orbitals, quantum numbers)
- Robert Mulliken – Molecular orbital theory
- Henry Eyring – Transition state theory (absolute reaction rates)
- Dorothy Crowfoot Hodgkin – X‑ray crystallography of complex molecules (penicillin, vitamin B12, insulin), Nobel 1964
- Glenn T. Seaborg – Transuranium elements (plutonium, americium, curium, etc.), actinide concept
- Frederick Sanger – First protein sequencing (insulin, 1955), DNA sequencing (Sanger method, 1977), two Nobel Prizes (1958, 1980)
- John Pople & Walter Kohn – Computational chemistry (density functional theory, DFT), Nobel 1998
- Richard R. Ernst & Kurt Wüthrich – NMR spectroscopy (Nobel 1991, 2002)
- Yves Chauvin, Robert Grubbs, Richard Schrock – Olefin metathesis (Nobel 2005)
- Osamu Shimomura, Martin Chalfie, Roger Y. Tsien – Green fluorescent protein (GFP, Nobel 2008)
- Dan Shechtman – Quasicrystals (Nobel 2011)
8. 21st Century to 2026: Modern Frontiers
- Click chemistry – Sharpless, Meldal, Bertozzi (Nobel 2022), bioorthogonal chemistry
- CRISPR‑Cas9 – Not chemistry alone but chemical biology applications (Nobel 2020 – Doudna, Charpentier, chemistry relevance)
- Lithium‑ion batteries – Goodenough, Whittingham, Yoshino (Nobel 2019)
- Machine learning in chemistry – Retrosynthesis prediction (2020–2026), property prediction, inverse design
- Single‑atom catalysis – Maximum atom efficiency (2010s–2026)
- Perovskite solar cells – Power conversion efficiency >25% (2026)
- MOFs (Metal‑Organic Frameworks) – Ultra‑high porosity, gas storage, separation (1990s–2026)
- Solid‑state batteries – Commercialization efforts (2020s)
- Plastic degradation enzymes – PETase (2020s)
- Green chemistry – 12 principles (Anastas & Warner, 1998), solvent‑free reactions, renewable feedstocks
- Flow chemistry – Continuous manufacturing
- Automated synthesis – Robot chemists (e.g., Chemputer)
Volume 2: Core Subdisciplines of Chemistry
9. Physical Chemistry
- Thermodynamics – Laws (0–3), Gibbs free energy (ΔG = ΔH – TΔS), enthalpy (H), entropy (S), Helmholtz free energy (A), chemical potential (μ), phase equilibria, Clapeyron & Clausius‑Clapeyron equations
- Kinetics – Reaction rates (rate = k[A]^m[B]^n), Arrhenius equation (k = A e^{−E_a/RT}), collision theory, transition state theory (ΔG‡), molecular dynamics (MD), potential energy surfaces
- Quantum chemistry – Schrödinger equation, Born‑Oppenheimer approximation, atomic orbitals (s, p, d, f), molecular orbitals (bonding/antibonding), Hartree‑Fock (HF), post‑HF (MP2, CCSD(T)), density functional theory (DFT – B3LYP, PBE0, M06), basis sets (STO‑3G, 6‑31G*, cc‑pVTZ), computational chemistry software (Gaussian, ORCA, Q‑Chem, NWChem)
- Statistical mechanics – Microstates, partition function (q), Boltzmann distribution, ensembles (microcanonical, canonical, grand canonical), ideal gas law from statistical mechanics, heat capacity
- Electrochemistry – Electrode potentials (Nernst equation, E = E° – (RT/nF) ln Q), standard reduction potentials (table), electrochemical cells (galvanic, electrolytic), Butler‑Volmer equation, cyclic voltammetry (CV), impedance spectroscopy
- Surface chemistry – Adsorption (Langmuir, BET), surface tension, catalysis (heterogeneous: active sites, Sabatier principle), surface science techniques (LEED, XPS, AES, STM, AFM)
10. Inorganic Chemistry
- Periodic table – Groups (1–18, IUPAC), periods, blocks (s, p, d, f), periodic trends (atomic radius, ionization energy, electron affinity, electronegativity, metallic character)
- Atomic structure – Quantum numbers (n, l, m_l, m_s), electron configurations (Aufbau, Pauli, Hund), shielding & effective nuclear charge (Z_eff)
- Chemical bonding – Ionic (electrostatic), covalent (Lewis structures, VSEPR, valence bond theory, hybridization), metallic (electron sea, band theory), coordination (dative covalent)
- Coordination chemistry – Ligands (monodentate, polydentate, chelate), coordination numbers (4, 6, 8), crystal field theory (CFT), ligand field theory (LFT), geometries (octahedral, tetrahedral, square planar), high‑spin vs. low‑spin, Jahn‑Teller distortion, spectrochemical series, isomerism (geometric, optical, linkage, coordination)
- Main group chemistry – Hydrogen (hydrides, H⁺, H⁻), alkali metals (high reactivity, crown ethers, cryptands), alkaline earth metals, boron (boranes, carboranes, boric acid), carbon (allotropes, organic), nitrogen (ammonia, hydrazine, nitric acid), oxygen (oxides, peroxides, superoxides), halogens (F₂ to At₂, interhalogens), noble gases (Xe compounds, XeF₄, XeO₃)
- Transition metals – d‑block, variable oxidation states, magnetic properties, colored complexes (d‑d transitions), catalytic activity (Pd, Pt, Rh, Ru, Ni, Fe, Co), metalloenzymes (Fe in hemoglobin, Mg in chlorophyll, Zn in carbonic anhydrase)
- Organometallic chemistry – Metal‑carbon bonds, 18‑electron rule, oxidative addition / reductive elimination, migratory insertion, β‑hydride elimination, homogeneous catalysis (Wilkinson’s catalyst, Grubbs catalyst for metathesis, palladium cross‑couplings: Suzuki, Heck, Sonogashira, Negishi, Stille), ferrocene (sandwich compound)
- Solid‑state chemistry – Crystal structures (cubic, hexagonal, tetragonal, etc.), Bravais lattices, unit cells, X‑ray diffraction (Bragg’s law), defects (vacancies, interstitials, Frenkel, Schottky), non‑stoichiometric compounds, superconductors (YBCO, cuprates), perovskites (ABX₃), zeolites (microporous aluminosilicates)
11. Organic Chemistry
- Hydrocarbons – Alkanes (CₙH₂ₙ₊₂, saturated), alkenes (CₙH₂ₙ, C=C), alkynes (CₙH₂ₙ₋₂, C≡C), cycloalkanes, aromatic (benzene, Hückel’s rule 4n+2)
- Functional groups – Alcohols (–OH), ethers (–O–), aldehydes (–CHO), ketones (–C=O), carboxylic acids (–COOH), esters (–COOR), amines (–NH₂, –NHR, –NR₂), amides (–CONH₂), nitriles (–CN), halides (–F, –Cl, –Br, –I), thiols (–SH), sulfides, sulfoxides, sulfones
- Stereochemistry – Chirality (asymmetric carbon), enantiomers, diastereomers, meso compounds, R/S nomenclature (Cahn‑Ingold‑Prelog), optical activity (polarimetry), racemic mixtures, resolution, chiral resolution, asymmetric synthesis (chiral catalysts, Sharpless epoxidation, Noyori hydrogenation)
- Reaction mechanisms – Nucleophilic substitution (S_N1, S_N2), elimination (E1, E2), addition (electrophilic, nucleophilic, free radical), rearrangement (carbocation, Wagner‑Meerwein), pericyclic (cycloaddition, electrocyclic, sigmatropic – Woodward‑Hoffmann rules)
- Named reactions (selection) – Grignard, Friedel‑Crafts (alkylation, acylation), Aldol, Claisen, Diels‑Alder, Wittig, Heck, Suzuki, Sonogashira, McMurry, Shapiro, Corey‑Fuchs, Pinnick oxidation, Swern oxidation, Baeyer‑Villiger, Beckmann, Hofmann, Curtius, Schmidt, Mannich, Michael, Robinson annulation, Stork enamine, Julia olefination, Peterson olefination, Appel, Mitsunobu, Ugi, Passerini, Biginelli
- Aromatic chemistry – Electrophilic aromatic substitution (nitration, halogenation, sulfonation, Friedel‑Crafts), nucleophilic aromatic substitution (S_NAr, benzyne mechanism), directing effects (ortho/para vs. meta), polycyclic aromatics (naphthalene, anthracene, pyrene)
- Heterocyclic chemistry – Pyrrole, furan, thiophene, pyridine, pyrimidine, purine (DNA bases), indole, quinoline, isoquinoline, azoles (imidazole, thiazole, oxazole)
- Natural products – Terpenes (isoprene units, monoterpenes to tetraterpenes), alkaloids (morphine, quinine, nicotine, caffeine), steroids (cholesterol, sex hormones, corticosteroids), polyketides, glycosides, flavonoids, lignans
- Polymer chemistry – Addition polymers (polyethylene, polypropylene, polystyrene, PVC, PTFE), condensation polymers (polyesters, polyamides – nylon, Kevlar), step‑growth vs. chain‑growth, living polymerization (ATRP, RAFT, NMP), cross‑linking, thermoplastics vs. thermosets, biopolymers (PLA, PHA), conductive polymers (polyaniline, PEDOT:PSS), polymer degradation
12. Analytical Chemistry
- Qualitative analysis – Spot tests, flame tests (Na⁺ yellow, K⁺ lilac, Cu²⁺ green), precipitation reactions, identification of functional groups (organic)
- Quantitative analysis – Gravimetric (mass), volumetric (titration: acid‑base, redox, complexometric, precipitation)
- Separation techniques – Distillation (simple, fractional, vacuum, steam), extraction (liquid‑liquid, solid‑phase – SPE), chromatography (TLC, column, GC, HPLC, UPLC), ion exchange, electrophoresis (gel, capillary), field‑flow fractionation
- Spectroscopy – UV‑Vis (π→π, n→π, Beer‑Lambert law), IR (vibrations: stretches, bends; fingerprint region, functional group identification), Raman (complementary to IR, water compatible), NMR (¹H, ¹³C, 2D – COSY, NOESY, HSQC, HMBC, DEPT), Mass spectrometry (EI, CI, ESI, MALDI, TOF, quadrupole, ion trap, orbitrap, FT‑ICR), Atomic spectroscopy (AA, AES, ICP‑MS)
- Electroanalytical methods – Potentiometry (ion‑selective electrodes, pH meter), conductometry, coulometry, voltammetry (CV, DPV, stripping voltammetry), amperometry
- Thermal analysis – TGA (thermogravimetric), DSC (differential scanning calorimetry), DTA
- Microscopy – Optical, SEM (scanning electron), TEM (transmission electron), AFM (atomic force), STM (scanning tunneling)
- Sensors & biosensors – Electrochemical (glucose sensor), optical (surface plasmon resonance – SPR), piezoelectric (quartz crystal microbalance – QCM)
13. Biochemistry (Interface with Chemistry)
- Proteins – Amino acids (20 standard), peptide bond, primary to quaternary structure, folding (hydrophobic effect, hydrogen bonds, disulfide bridges), enzymes (catalysis, Michaelis‑Menten kinetics, K_m, V_max, inhibition types)
- Nucleic acids – DNA (double helix, A‑T, G‑C base pairs), RNA (mRNA, tRNA, rRNA, non‑coding), replication, transcription, translation, genetic code, mutations
- Carbohydrates – Monosaccharides (glucose, fructose), disaccharides (sucrose, lactose), polysaccharides (starch, glycogen, cellulose, chitin), glycosidic bonds
- Lipids – Fatty acids (saturated, unsaturated), triglycerides, phospholipids (membrane bilayers), steroids, terpenes
- Metabolism – Glycolysis, TCA cycle (Krebs), oxidative phosphorylation (ETC, ATP synthase), gluconeogenesis, fatty acid oxidation (β‑oxidation), photosynthesis (light reactions, Calvin cycle)
- Chemical biology – Bioorthogonal chemistry (click, tetrazine ligation), activity‑based probes, chemical genetics, optogenetics
14. Materials Chemistry
- Ceramics – Oxides (Al₂O₃, ZrO₂), nitrides (Si₃N₄), carbides (SiC, WC), glasses (SiO₂‑based), glazes
- Metals & alloys – Steel (Fe + C), stainless steel (Cr, Ni), brass (Cu+Zn), bronze (Cu+Sn), superalloys (Ni‑based), shape‑memory alloys (Nitinol)
- Semiconductors – Si, Ge, GaAs, GaN, SiC, doping (n‑type, p‑type), band gap engineering
- Nanomaterials – Nanoparticles (Au, Ag, Pt, quantum dots – CdSe, PbS), nanotubes (carbon, BN), nanowires, graphene (single layer of graphite), 2D materials (MoS₂, WS₂, MXenes), fullerenes (C₆₀, C₇₀)
- Polymers (see organic chemistry above)
- Composites – Carbon fiber reinforced polymers (CFRP), glass fiber reinforced polymers (GFRP), ceramic matrix composites (CMC)
- Biomaterials – Hydrogels, biodegradable polymers (PLGA, PCL), bone cements (PMMA), dental composites, artificial skin
- Porous materials – Zeolites, MOFs (metal‑organic frameworks), COFs (covalent organic frameworks), mesoporous silica (SBA‑15, MCM‑41)
15. Nuclear Chemistry
- Radioactivity – Alpha (α, ⁴He), beta (β⁻, e⁻; β⁺, positron), gamma (γ, high energy photon), neutron emission, spontaneous fission
- Decay kinetics – Half‑life (t₁/₂), activity (A = λN), decay series (U‑238, Th‑232, U‑235)
- Nuclear reactions – Fusion (light nuclei combine, e.g., p‑p chain in Sun), fission (heavy nuclei split, e.g., U‑235 + n → fission products + 2‑3 n + energy)
- Radiochemical methods – Neutron activation analysis (NAA), isotope dilution, radiometric dating (¹⁴C, ⁴⁰K‑⁴⁰Ar, U‑Pb, Rb‑Sr)
- Applications – Nuclear power (fission reactors, Breeder reactors, fusion research – ITER), nuclear medicine (PET, SPECT, radiotherapy, ⁹⁹ᵐTc), industrial tracers, smoke detectors (²⁴¹Am)
16. Environmental & Green Chemistry
- Atmospheric chemistry – Ozone layer (CFCs, O₃ depletion, Montreal Protocol), greenhouse gases (CO₂, CH₄, N₂O, SF₆), photochemical smog (NOₓ, VOCs, PAN), acid rain (SO₂, NOₓ → H₂SO₄, HNO₃)
- Water chemistry – Hardness (Ca²⁺, Mg²⁺), wastewater treatment (primary, secondary – activated sludge, tertiary), eutrophication (phosphate, nitrate)
- Soil chemistry – pH buffering, cation exchange capacity (CEC), heavy metals (Pb, Hg, Cd, Cr), pesticides, fertilizers
- Green chemistry principles – 12 principles (prevention, atom economy, less hazardous synthesis, safer solvents, renewable feedstocks, catalysis, design for degradation, real‑time analysis, accident prevention)
- Sustainable chemistry – CO₂ capture (amine scrubbing, MOFs), renewable feedstocks (biomass, waste), plastic recycling (mechanical, chemical – depolymerization, pyrolysis), biodegradable polymers
Volume 3: Special Topics & Frontiers (up to 2026)
17. Supramolecular Chemistry
- Non‑covalent interactions – Hydrogen bonding, π‑π stacking, van der Waals, hydrophobic effect, electrostatic, halogen bonding
- Host‑guest chemistry – Crown ethers (pedersen), cryptands (lehn), cyclodextrins, calixarenes, cucurbiturils
- Self‑assembly – Micelles, vesicles, Langmuir‑Blodgett films, DNA origami, metal‑organic polyhedra (MOPs)
- Molecular machines – Rotaxanes, catenanes, molecular motors (Sauvage, Stoddart, Feringa, Nobel 2016), molecular switches
18. Click Chemistry & Bioorthogonal Chemistry
- Click reactions – CuAAC (copper‑catalyzed azide‑alkyne cycloaddition), SPAAC (strain‑promoted, no Cu), Diels‑Alder, thiol‑ene
- Bioorthogonal ligations – Tetrazine‑trans‑cyclooctene (TCO), azide‑phosphine (Staudinger ligation)
- Applications – Drug conjugation (ADC), biomolecule labeling (in vivo, live cells), materials functionalization
19. Catalysis (Homogeneous, Heterogeneous, Biocatalysis)
- Homogeneous – Organometallic complexes (Pd, Rh, Ru), chiral catalysts (asymmetric hydrogenation – Noyori, Knowles; epoxidation – Sharpless; dihydroxylation – Sharpless)
- Heterogeneous – Supported metals (Pt, Pd on carbon, zeolites), metal oxides (V₂O₅, TiO₂), single‑atom catalysts (SACs, 2010s–2026)
- Biocatalysis – Enzymes (lipases, proteases, aldolases, transaminases), engineered enzymes (directed evolution – Arnold, Nobel 2018)
- Photocatalysis – TiO₂ (UV), visible‑light photocatalysts (Ru(bpy)₃²⁺, Eosin Y), organic photoredox
- Electrocatalysis – Pt for HER (hydrogen evolution), IrO₂ for OER (oxygen evolution), Fe‑N‑C for ORR (oxygen reduction), CO₂ reduction catalysts (Cu, Au, Ag, molecular catalysts)
20. Computational Chemistry & Cheminformatics
- Molecular modeling – Molecular mechanics (force fields: MMFF, AMBER, CHARMM, OPLS), MD simulations (GROMACS, NAMD, AMBER), Monte Carlo (MC)
- Electronic structure – HF, DFT (B3LYP, PBE, M06‑2X, ωB97X‑D), post‑HF (MP2, CCSD(T)), basis sets, pseudopotentials
- Chemoinformatics – SMILES, InChI, molecular fingerprints (Morgan, ECFP), QSAR (quantitative structure‑activity relationship), virtual screening, molecular docking (AutoDock, Glide)
- Machine learning in chemistry – Neural network potentials (ANI, SchNet, GNNs), property prediction (logP, solubility, toxicity), retrosynthesis prediction (2020–2026 tools: IBM RXN, ASKCOS, AiZynthFinder), inverse design (generative models – VAEs, GANs, diffusion)
21. Electrochemistry & Energy Storage
- Batteries – Lead‑acid, Ni‑MH, Li‑ion (intercalation cathodes: LiCoO₂, NMC, LFP; anodes: graphite, silicon, Li metal), solid‑state batteries (sulfide, oxide electrolytes), Li‑S, Li‑air, Na‑ion, K‑ion, multivalent (Mg, Zn, Al)
- Supercapacitors – Double‑layer capacitance (EDLC – activated carbon), pseudocapacitance (metal oxides, conductive polymers)
- Fuel cells – PEMFC (proton exchange membrane – H₂/air, Pt catalyst), SOFC (solid oxide, high temperature), DMFC (direct methanol)
- Water splitting – HER (cathode), OER (anode), bifunctional catalysts, photoelectrochemical (PEC) cells
- CO₂ reduction – Electrochemical CO₂RR to CO, formate, methane, ethylene, ethanol (Cu based)
22. Photochemistry & Artificial Photosynthesis
- Jablonski diagram – Singlet (S₀, S₁, S₂), triplet (T₁), fluorescence (ns), phosphorescence (μs–s), intersystem crossing, internal conversion
- Organic photovoltaics (OPVs) – Bulk heterojunction (donor: polymer, acceptor: PCBM, non‑fullerene acceptors – Y6), power conversion efficiency >19% (2026)
- Dye‑sensitized solar cells (DSSCs) – TiO₂, Ru dyes (N719, N3), electrolyte (I⁻/I₃⁻), efficiency ~12%
- Perovskite solar cells – MAPbI₃, FAPbI₃, efficiency >25% (2026), stability improvements, tandem cells (perovskite/Si >30%)
- Artificial photosynthesis – Water oxidation catalysts, CO₂ reduction catalysts, photocatalytic N₂ fixation (NH₃)
23. Chemical Biology & Drug Discovery
- Hit‑to‑lead – High‑throughput screening (HTS), fragment‑based drug design (FBDD), DNA‑encoded libraries (DEL)
- Structure‑based design – X‑ray crystallography, cryo‑EM, NMR, molecular docking
- Pharmacokinetics (ADME) – Absorption, distribution, metabolism, excretion, toxicity (ADMET)
- Small molecule drugs – Antibiotics (penicillins, cephalosporins, macrolides, quinolones, tetracyclines), antivirals (paxlovid, remdesivir, antiretrovirals), anticancer agents (kinase inhibitors – imatinib, osimertinib; PARP inhibitors, PROTACs)
- Biologics – Monoclonal antibodies (mAbs – adalimumab, pembrolizumab), antibody‑drug conjugates (ADCs – trastuzumab emtansine), peptides, oligonucleotides (ASOs, siRNA – inclisiran), mRNA drugs (COVID vaccines)
- Chemical proteomics – Activity‑based protein profiling (ABPP), affinity pulldowns, thermal shift assays (CETSA)
Volume 4: Major Industrial & Applied Chemistry
24. Industrial Processes (Large‑Scale)
- Haber‑Bosch – N₂ + 3 H₂ ⇌ 2 NH₃ (Fe catalyst, high pressure, high temperature, ~200 bar, 400–500°C)
- Contact process – SO₂ + ½ O₂ → SO₃ (V₂O₅ catalyst) → H₂SO₄
- Ostwald process – NH₃ → HNO₃ (Pt/Rh gauze)
- Chlor‑alkali – Electrolysis of brine (NaCl) → Cl₂, H₂, NaOH
- Cracking & refining – Fluid catalytic cracking (FCC), steam cracking (ethylene, propylene), catalytic reforming, alkylation
- Fischer‑Tropsch – CO + H₂ → liquid hydrocarbons (Fe or Co catalyst, from coal/gas/biomass)
- Methanol synthesis – CO + 2 H₂ → CH₃OH (Cu/ZnO/Al₂O₃)
- Cement production – Limestone (CaCO₃) → CaO + CO₂, clinker formation
- Steelmaking – Blast furnace (iron ore → pig iron), basic oxygen furnace (BOF), electric arc furnace (EAF)
- Polymers – Ziegler‑Natta polymerization (polyethylene, polypropylene), metallocene catalysis, PET production (terephthalic acid + ethylene glycol), nylon (hexamethylenediamine + adipic acid)
25. Analytical Instrumentation (Modern)
- Mass spectrometry – High resolution (Orbitrap, FT‑ICR), tandem MS (MS/MS), imaging MS (MALDI‑IMS)
- NMR – 1+ GHz spectrometers, cryoprobes, solid‑state NMR (MAS), benchtop NMR (2020s)
- Crystallography – X‑ray free electron lasers (XFEL), serial crystallography (room temperature, time‑resolved)
- Electron microscopy – Cryo‑EM (single particle analysis, Nobel 2017), cryo‑electron tomography (CET)
- Single‑molecule methods – Optical tweezers, AFM, scanning probe, super‑resolution microscopy (STED, PALM, STORM)
26. Safety, Hazards & Chemical Regulation
- GHS (Globally Harmonized System) – Hazard pictograms (9 symbols), H‑phrases, P‑phrases
- REACH (EU, 2007) – Registration, Evaluation, Authorisation, Restriction of Chemicals
- TSCA (US Toxic Substances Control Act)
- Chemical safety – Flammability (flash point, autoignition), toxicity (LD₅₀, LC₅₀), carcinogenicity (IARC classes), corrosivity, reactivity (peroxides, water‑reactive, pyrophorics)
- Personal protective equipment (PPE) – Gloves (nitrile, neoprene, butyl), goggles, faceshield, lab coat, fume hood, blast shield
- Waste disposal – Hazardous waste, organic solvents, heavy metals, sharps, radioactive waste
Volume 5: People, Institutions & Prizes
27. Notable Chemists (Biographical entries – selection)
- Lavoisier, Dalton, Berzelius, Mendeleev, Curie (Marie & Pierre), Pauling, Hodgkin, Seaborg, Sanger, Sharpless, Bertozzi, Goodenough, Whittingham, Yoshino, Meldal, Doudna, Charpentier, Arnold, Hoffmann (Roald), Zewail (femtochemistry), Eigen (flash photolysis), Pople, Kohn, van ’t Hoff, Arrhenius, Ostwald, Fischer (Emil), Baeyer, Liebig, Wöhler, Linus Pauling, Robert H. Grubbs, Richard R. Schrock, Yves Chauvin, Alan Heeger (conducting polymers), Hideki Shirakawa, Alan MacDiarmid
28. Major Chemical Societies & Institutions
- American Chemical Society (ACS) – Largest scientific society (founded 1876)
- Royal Society of Chemistry (RSC) (UK)
- Gesellschaft Deutscher Chemiker (GDCh) (Germany)
- Chemical Society of Japan (CSJ)
- International Union of Pure and Applied Chemistry (IUPAC) – Standardizes nomenclature, atomic weights, periodic table
- European Federation of Chemical Engineering (EFCE)
- Nobel Prize in Chemistry (since 1901)
- Wolf Prize in Chemistry
- Japan Prize
- ACS Award in Pure Chemistry
29. Nobel Prizes in Chemistry (Selected Landmarks 1901–2025)
- 1901 – van ’t Hoff (chemical dynamics, osmotic pressure)
- 1909 – Ostwald (catalysis, chemical equilibria)
- 1911 – Curie (radium, polonium)
- 1915 – Willstätter (chlorophyll)
- 1918 – Haber (ammonia synthesis)
- 1920 – Nernst (thermodynamics)
- 1921 – Soddy (isotopes, radioactive decay)
- 1922 – Aston (mass spectrometry)
- 1926 – Svedberg (ultracentrifuge)
- 1928 – Windaus (sterols, vitamin D)
- 1932 – Langmuir (surface chemistry)
- 1934 – Urey (deuterium)
- 1935 – Joliot‑Curie (artificial radioactivity)
- 1938 – Kuhn (carotenoids, vitamins)
- 1944 – Hahn (fission)
- 1945 – Virtanen (agricultural chemistry)
- 1946 – Sumner (enzyme crystallization), Northrop, Stanley (virus purification)
- 1947 – Robinson (alkaloids)
- 1949 – Giauque (low‑temperature thermodynamics)
- 1951 – McMillan, Seaborg (transuranium)
- 1954 – Pauling (chemical bond)
- 1958 – Sanger (insulin)
- 1962 – Perutz, Kendrew (hemoglobin, myoglobin)
- 1963 – Ziegler, Natta (polymerization)
- 1964 – Hodgkin (vitamin B12)
- 1965 – Woodward (organic synthesis)
- 1970 – Lel
Cross-referenced network for Sarvarthapedia
Chemistry → See also: Atomic Structure, Periodic Table, Chemical Reactions, Physical Chemistry, Organic Chemistry, Biochemistry, Materials Chemistry, Thermodynamics, Chemical Kinetics, Quantum Chemistry, Environmental Chemistry, Industrial Chemistry, Computational Chemistry, Universe Chemistry, Human Chemical Composition, Indian Chemistry
Atomic Structure → See also: Subatomic Particles, Electron Configuration, Quantum Mechanics, Quantum Chemistry, Periodic Table, Chemical Bonding, Nuclear Chemistry
Subatomic Particles → See also: Protons, Neutrons, Electrons, Atomic Structure, Nuclear Chemistry, Quantum Mechanics
Quantum Mechanics → See also: Wave Functions, Energy Levels, Orbitals, Quantum Chemistry, Physical Chemistry, Atomic Structure
Periodic Table → See also: Chemical Elements, Atomic Number, Periodicity, Electron Configuration, Chemical Bonding, Inorganic Chemistry
Chemical Elements → See also: Periodic Table, Atomic Structure, Isotopes, Chemical Bonding, Universe Chemistry
Chemical Bonding → See also: Ionic Bonds, Covalent Bonds, Metallic Bonds, Molecular Structure, Organic Chemistry, Materials Chemistry
Molecular Structure → See also: Chemical Bonding, Organic Chemistry, Biochemistry, Spectroscopy, Quantum Chemistry
Chemical Reactions → See also: Reaction Mechanisms, Thermodynamics, Chemical Kinetics, Catalysis, Equilibrium, Biochemistry
Reaction Mechanisms → See also: Chemical Reactions, Catalysis, Organic Chemistry, Kinetics
Thermodynamics → See also: Enthalpy, Entropy, Gibbs Free Energy, Energy Transfer, Physical Chemistry, Chemical Reactions
Chemical Kinetics → See also: Reaction Rates, Activation Energy, Catalysts, Equilibrium, Thermodynamics
Catalysis → See also: Enzymes, Reaction Mechanisms, Industrial Chemistry, Biochemistry
Equilibrium → See also: Chemical Reactions, Thermodynamics, Le Chatelier Principle, Kinetics
Physical Chemistry → See also: Thermodynamics, Chemical Kinetics, Quantum Chemistry, Energy Systems, Atomic Structure
Organic Chemistry → See also: Carbon Compounds, Hydrocarbons, Polymers, Pharmaceuticals, Biochemistry, Reaction Mechanisms
Carbon Compounds → See also: Organic Chemistry, Biochemistry, Molecular Structure, Materials Chemistry
Biochemistry → See also: Nucleic Acids, Proteins, Enzymes, Metabolism, Molecular Biology, Organic Chemistry
Nucleic Acids → See also: DNA, RNA, Genetic Information, Biochemistry, Molecular Biology
Proteins → See also: Enzymes, Amino Acids, Biochemistry, Metabolism
Enzymes → See also: Catalysis, Proteins, Biochemistry, Metabolism
Metabolism → See also: Biochemistry, Energy Transfer, Enzymes, Human Chemical Composition
Materials Chemistry → See also: Nanotechnology, Semiconductors, Biomaterials, Solid State Chemistry, Industrial Chemistry
Nanotechnology → See also: Materials Chemistry, Quantum Chemistry, Computational Chemistry, Biotechnology
Industrial Chemistry → See also: Commercial Chemistry, Manufacturing, Catalysis, Green Chemistry, Materials Chemistry
Commercial Chemistry → See also: Industrial Chemistry, Pharmaceuticals, Agriculture, Energy, Consumer Products
Green Chemistry → See also: Sustainability, Environmental Chemistry, Industrial Chemistry, Waste Reduction
Environmental Chemistry → See also: Pollution, Climate Science, Ecosystems, Green Chemistry
Computational Chemistry → See also: Artificial Intelligence, Molecular Modeling, Quantum Chemistry, Nanotechnology
Artificial Intelligence → See also: Computational Chemistry, Data Science, Automation, Molecular Simulation
Universe Chemistry → See also: Big Bang, Stellar Nucleosynthesis, Planetary Formation, Chemical Elements
Big Bang → See also: Universe Chemistry, Cosmology, Particle Physics
Stellar Nucleosynthesis → See also: Universe Chemistry, Chemical Elements, Astrophysics
Planetary Formation → See also: Universe Chemistry, Geochemistry, Atmospheres
Human Chemical Composition → See also: Biochemistry, Metabolism, Homeostasis, Molecular Biology
Homeostasis → See also: Human Chemical Composition, Biochemistry, Physiology
Indian Chemistry → See also: Ayurveda, Rasashastra, Metallurgy, Modern Chemical Research
Ayurveda → See also: Indian Chemistry, Herbal Compounds, Biochemistry
Rasashastra → See also: Indian Chemistry, Alchemy, Metallurgy
Metallurgy → See also: Materials Chemistry, Industrial Chemistry, Rasashastra
Modern Chemical Research → See also: Computational Chemistry, Nanotechnology, Research Institutions
Research Institutions → See also: Scientific Research, Innovation, Interdisciplinary Science
Core Concept: Chemistry
See also: Atomic Structure; Periodic Table; Chemical Reactions; Physical Chemistry; Organic Chemistry; Materials Chemistry; Biochemistry; Nucleic Acids; Universe Chemistry; Human Chemical Composition; Commercial Chemistry; Military Chemistry; Indian Chemistry; Research Institutions; Green Chemistry; Computational Chemistry — Chemistry serves as the central node in the Sarvarthapedia conceptual network, linking the study of matter and its transformations to both the physical sciences and life sciences, while integrating fundamental principles such as atomic structure and quantum mechanics with applied domains like industrial production, biological systems, and cosmic evolution, thereby creating a unified framework in which all material phenomena are interconnected.
Core Concept: Atomic Structure
See also: Quantum Mechanics; Electron Orbitals; Periodic Table; Chemical Bonding; Chemical Reactions; Physical Chemistry — Atomic Structure forms the foundational layer of chemistry, explaining how protons, neutrons, and electrons are arranged, and linking directly to Periodic Table organization and chemical bonding, while also connecting to quantum theory and influencing all subsequent chemical interactions and transformations.
Core Concept: Periodic Table
See also: Atomic Structure; Chemical Bonding; Periodicity; Elements; Inorganic Chemistry — The Periodic Table organizes elements based on atomic number and electron configuration, establishing relationships such as electronegativity and ionization energy, and acting as a bridge between atomic theory and chemical reactivity across all branches of chemistry.
Core Concept: Chemical Reactions
See also: Chemical Bonding; Thermodynamics; Chemical Kinetics; Catalysis; Biochemistry — Chemical Reactions describe the transformation of substances through bond formation and breaking, connecting fundamental laws like conservation of mass with applied processes in industry, biology, and environmental systems, while being governed by energy changes and reaction rates.
Core Concept: Physical Chemistry
See also: Thermodynamics; Quantum Mechanics; Chemical Kinetics; Energy Systems — Physical Chemistry integrates physics principles into chemical systems, linking atomic-level interactions with macroscopic properties, and serving as a theoretical backbone for understanding reaction mechanisms and energy transformations.
Core Concept: Organic Chemistry
See also: Carbon Compounds; Biochemistry; Polymers; Pharmaceuticals; Materials Chemistry — Organic Chemistry focuses on carbon-based molecules, forming a conceptual bridge between chemical structure and biological function, while also connecting to industrial applications such as drug development and synthetic materials.
Core Concept: Materials Chemistry
See also: Nanotechnology; Semiconductors; Biomaterials; Industrial Chemistry — Materials Chemistry links chemical synthesis with engineering applications, focusing on designing substances with specific mechanical, electrical, and biological properties, thereby connecting fundamental chemistry with technology and innovation.
Core Concept: Biochemistry
See also: Nucleic Acids; Proteins; Metabolism; Molecular Biology — Biochemistry explores the chemical basis of life, connecting organic chemistry with biological systems, and demonstrating how chemical reactions underpin processes such as metabolism, genetic replication, and cellular function.
Core Concept: Nucleic Acids
See also: DNA; RNA; Genetic Information; Biochemistry; Molecular Biology — Nucleic Acids act as carriers of genetic information, linking chemical structure with biological inheritance, and forming a crucial intersection between chemistry and life sciences.
Core Concept: Human Chemical Composition
See also: Biochemistry; Metabolism; Homeostasis; Life Processes — Human beings are understood as complex assemblies of chemical compounds, where life emerges from dynamic molecular interactions, linking philosophical interpretations of existence with scientific explanations rooted in chemistry.
Core Concept: Universe Chemistry
See also: Big Bang; Stellar Nucleosynthesis; Planetary Formation; Astrophysics — Universe Chemistry connects the origin of matter from the Big Bang to the formation of stars and planets, illustrating how chemical elements evolve through cosmic processes, thereby linking chemistry with astronomy and cosmology.
Core Concept: Commercial Chemistry
See also: Industrial Chemistry; Pharmaceuticals; Agriculture; Energy; Green Chemistry — Commercial Chemistry highlights the application of chemical principles in economic activities, connecting laboratory discoveries to mass production, consumer goods, and sustainable technologies.
Core Concept: Military Chemistry
See also: Explosives; Chemical Weapons; Ethics in Science; International Regulation — Military Chemistry represents the application of chemical knowledge in defense and warfare, linking scientific advancement with ethical considerations and global policy frameworks.
Core Concept: Research Institutions
See also: Scientific Innovation; Interdisciplinary Research; Global Collaboration — Research Institutions act as hubs for chemical advancement, connecting education, experimentation, and technological development, while fostering integration across physics, biology, and engineering.
Core Concept: Computational and AI Chemistry
See also: Artificial Intelligence; Molecular Modeling; Nanotechnology; Materials Chemistry — Computational Chemistry integrates digital technologies with chemical research, enabling simulation of molecular systems and accelerating discoveries in materials science and drug design.
Core Concept: Indian Chemistry
See also: Ayurveda; Rasashastra; Metallurgy; Modern Chemical Research; Global Science — Indian Chemistry forms a continuous knowledge system linking ancient practices such as Ayurveda and alchemy with modern scientific research, integrating traditional wisdom with contemporary advancements in materials science, biochemistry, and nanotechnology, thereby connecting regional knowledge to the global chemical network.
Core Concept: Green Chemistry
See also: Sustainability; Environmental Chemistry; Industrial Processes — Green Chemistry connects chemical innovation with environmental responsibility, emphasizing the design of processes that reduce waste, minimize toxicity, and promote sustainable development, linking science with global ecological concerns.